Germfree will create a suite of cGMP documents that can be used by the client to provide objective evidence to a regulatory body (CDC, FDA, etc) that the facility or equipment was built and operates as designed and meets the client User Requirements Specification (URS). These documents include a commissioning plan, FAT/SAT protocols, and a summary report.
Innovative CQV Solutions

Full Commissioning Process
We will take care of everything from user requirement specs, FAT/SAT testing, to IOQ and turnover packages.
Highly Versed Team
Our CQV Engineers are not only specialized in testing our mobile and modular technology, but are also highly versed with the regulatory requirements of the pharmaceutical, medical device, and biocontainment industries.
Onsite Team
Our expert team is located at our manufacturing factory allowing full access to the facility and other Germfree resources from the early design phase through product completion. This allows CQV documents and protocols to be highly detailed and custom tailored to each project.
Collaborative Approach
Client involvement is encouraged during FAT and SAT executions which provides a collaborative approach and ensures thorough and unbiased testing.

Planning for Commissioning
In collaboration with the client, Germfree will prepare the commissioning plan for the equipment, mobile, or modular facility. The plan outlines the scope, tasks, procedures, roles, and responsibilities of various involved groups, and the overall coordination of the commissioning effort.
Our Process
Our CQV process contains a series of detailed tests and supporting evidence that the equipment or facility’s system meets requirements and is executed at our factory following good documentation practices.
Factory Acceptance Testing (FAT)
Commissioning at this phase assures that the facility meets customer/design requirements prior to shipment to its final destination. At the factory, every attempt will be made to simulate a completed facility to provide the commissioning team the opportunity to test the facility and systems to the fullest extent possible. This approach minimizes encountering issues onsite and helps reduce site installation time thus expediting the site acceptance testing phase of the project.
Site Acceptance Testing (SAT)
Commissioning at the final destination utilizes a risk-based approach allowing the leveraging of non-critical tests previously performed at the factory and therefore reducing the amount of testing performed on site. This will ensure the facility meets customer/design requirements in its final state with the production build complete and facilitates project completion and turnover.
Installation and Operational Qualification (IOQ)
We will author and execute a Germfree Cleanroom Facility testing protocol providing objective evidence that the cleanroom meets both clean air (ISO14644) and FDA-cGMP requirements. This includes materials of construction, temperature, humidity, and differential pressure trending, one day static, and non-viable particulate studies. The manufacturer of the EMS system will execute their installation and operational qualification (vendor protocol) following installation completion. Finally, we will execute an IOQ protocol for various equipment such as: pass-thru chambers, restrictive access barrier (RABS), gloveboxes and containment systems and provide a summary report.
Turnover Package
At the end of the commissioning process, we will prepare a turnover package that provides a central documentation reference for all systems. This package consists of a combination of documents provided by Germfree and their subcontractors. It also includes a narrative summary report of the commissioning process summarizing key information, deficiencies, open issues (if any), and overall results.
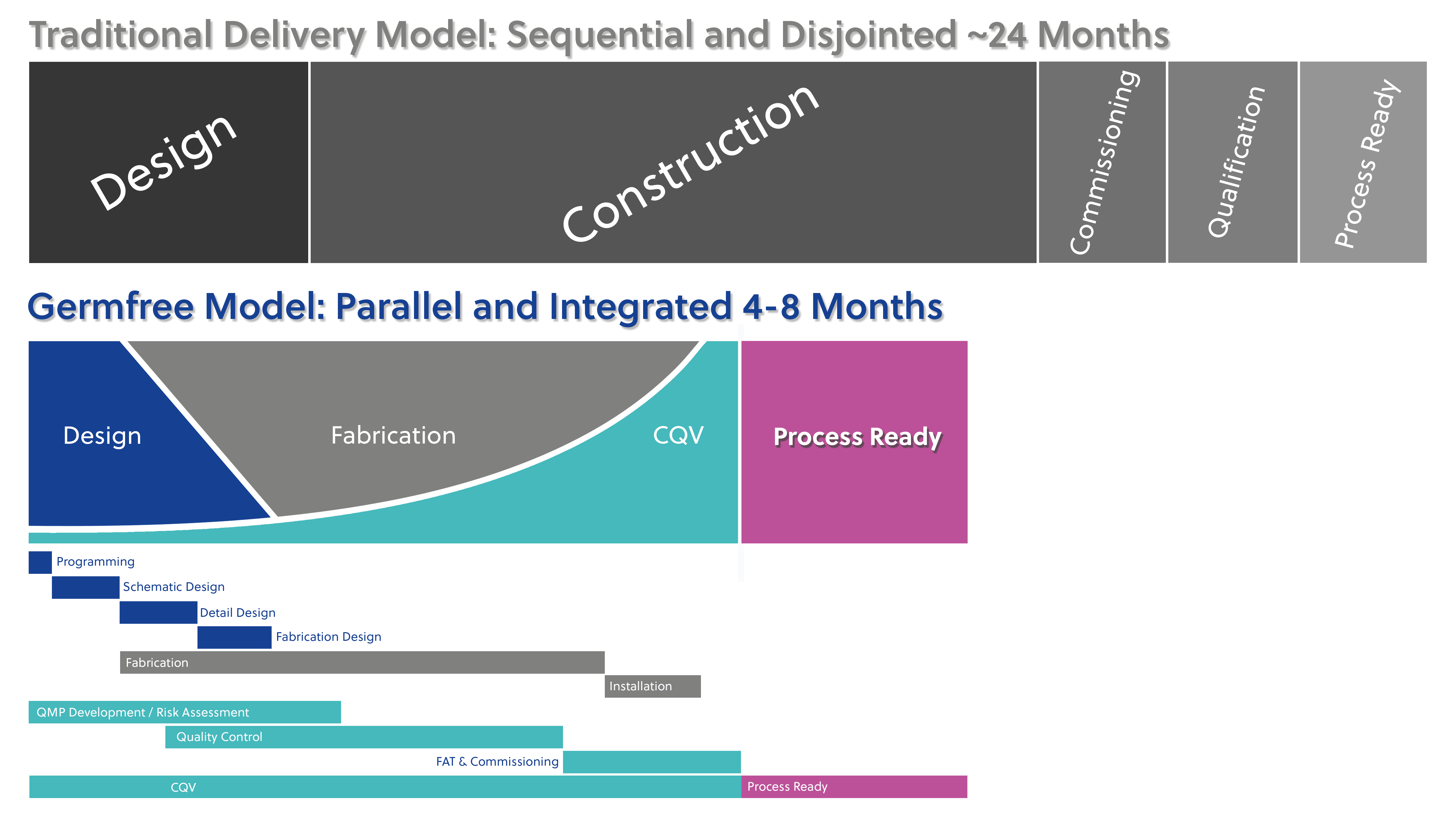
Reduced Time To Process Ready
Our CQV services cover all stages of your facility’s lifecycle, from design and construction to operation and maintenance. With a team of highly trained and innovative professionals, we help clients minimize risk, increase efficiency, and maintain compliance, ultimately enabling them to bring safe and effective products to market.
Let's build something.
Tell us about your project and we’ll connect you with the right team within 24 hours.